


The human body is so intricate that a single medical mistake, however small, can have devastating effects.
Medical errors happen with unthinkable frequency in the United States. If your family has suffered a severe injury at the hands of a negligent doctor, it’s essential that you get help right away to cope with the shattering effects of malpractice: pain and disability, astronomical medical bills, or even – for hundreds of thousands of families – the irrevocable loss of a loved one’s life.
It might feel like there’s nothing anyone can do to make things better. The medical mistake can’t be undone, and neither can the consequences it brought upon your health and your family.
But it doesn’t have to be this way.
As a victim of medical malpractice, it’s understandable that you’re hesitant to put your faith in someone else – even a professional. After all, the last time you trusted a medical provider, the mistake they made cost you everything. It can make putting control of any part of your life in the hands of a stranger feel dangerous – like you’re inviting even more uncertainty into your life.
Here are the things you can feel certain of when you move forward with a medical malpractice claim with Console & Associates on your side:
Call (856) 778-5500 or send a secure online message to our medical malpractice lawyers right now. You pay no legal fees unless we win money for you – guaranteed.
A patient should never have to worry about whether they can trust their doctors, but the truth is that medical negligence happens all too often. If you suspect that something went wrong during your treatment, the personal injury attorneys at Console & Associates, P.C. want to hear from you right away. We can help you determine if your injury was the result of medical negligence and, if so, start building your case today.
If you’re looking for answers about your care or searching for a solution to the financial problems caused by the medical error, we’re ready to assist you. Drawing upon our decades of experience handling medical malpractice claims, we can help you fight back, get justice, and get compensation for all that you have been through.
Medical malpractice cases can be complicated, but we’ll walk you through every step of the process so that pursuing a claim doesn’t feel overwhelming. Our team of attorneys has recovered numerous medical malpractice negligence settlements on behalf of our clients—and we never charge upfront fees for our services. You can trust us to handle your case with care and compassion and to have your back during this difficult time.
You don’t have to go through this alone! Call us today at (866) 778-5500, and let our medical malpractice attorneys be your advocates.

Clipboard With Documents About Medical Malpractice And Gavel.
If you believe you may be the victim of a medical professional’s negligent actions, there’s no time to lose.
If the medical malpractice is ongoing or if your injuries put your health in immediate danger, you need to get to a safe place where you can be taken care of appropriately.
That may mean medical negligence or getting transferred to a different facility or a different physician.
The evidence of your doctor’s negligence is encapsulated in your medical charts. The sooner you are able to get your hands on these records, the sooner you can begin moving forward with your claim.
Patients have a legal right to their medical records. But the medical providers and facilities where negligence occurs aren’t always forthcoming. They may try to deter you from retrieving your medical records or make it difficult to access your full records in an attempt to keep medical mistakes hidden.
When you need to gather your medical records, you should start by presenting a written request to the medical facility or health system.
The United States Department of Health and Human Services ‘Privacy Rule’ outlines your rights with regard to accessing your medical records and ensuring they are kept private according to HIPPA law.
For a successful medical malpractice claim, you’re going to need a knowledgeable attorney on your side. All it takes to launch an investigation into your medical care is a simple phone call to a medical malpractice lawyer in New Jersey.
NJ medical negligence attorneys typically offer free consultations and will review your medical records – with the help of experts in the medical field – at no charge. Through this thorough review and investigation, we can determine whether you have medical malpractice grounds for a viable case.
Pursuing a medical malpractice and personal injury claim is crucial to your recovery and wellbeing. An insurance claim or lawsuit will be your only option to get compensation for the injuries you suffered due to the medical error and for the ways this injury has affected your life.
However, you also have the option to file a medical malpractice report with the state.
Who to report medical malpractice to in NJ:
New Jersey Department of Health Division of Health Facilities Evaluation and Licensing P.O. Box 367 Trenton NJ 08625-0367
Download the official State Board of Medical Examiners complaint form and mail to: Division of Consumer Affairs State Board of Medical Examiners P.O. Box 183 Trenton, NJ 08625
Download the official New Jersey Board of Nursing complaint form and mail to: New Jersey Board of Nursing P.O. Box 45010 Newark, NJ 07101
Medical errors take so many different forms that the signs of medical malpractice can differ significantly from one case to another. A few of the possible “red flags” of medical malpractice include:
Some medical malpractice claims involve more than one of these signs, while others are unique circumstances in which none of these signs apply.
If you have any reason to believe you or a loved one may have been a victim of medical malpractice, don’t hesitate to contact a personal injury attorney in New Jersey for assistance. It costs nothing to find out your options and nothing upfront to pursue a claim.
Dangerously poor treatment at the hands of a medical professional isn’t something you have to put up with. If you suffered serious injury as a direct result of a doctor’s negligence, you can file a medical malpractice insurance claim or a lawsuit. Through this legal action, you can:
For many victims of medical malpractice, the idea of suing anyone can seem daunting. When the defendant you’re suing is a doctor you once trusted or a large hospital or healthcare system, the prospect of a lawsuit may be even more intimidating.
Here are four common concerns – and why you can’t afford to let these fears stand in the way of moving forward with your claim.
1. Fear of not being able to afford the cost of a medical malpractice lawyer
You may be afraid you’ll have to pay a high price to talk to an attorney.
It’s a myth that hiring New Jersey medical malpractice attorneys is unaffordable – but a common one.
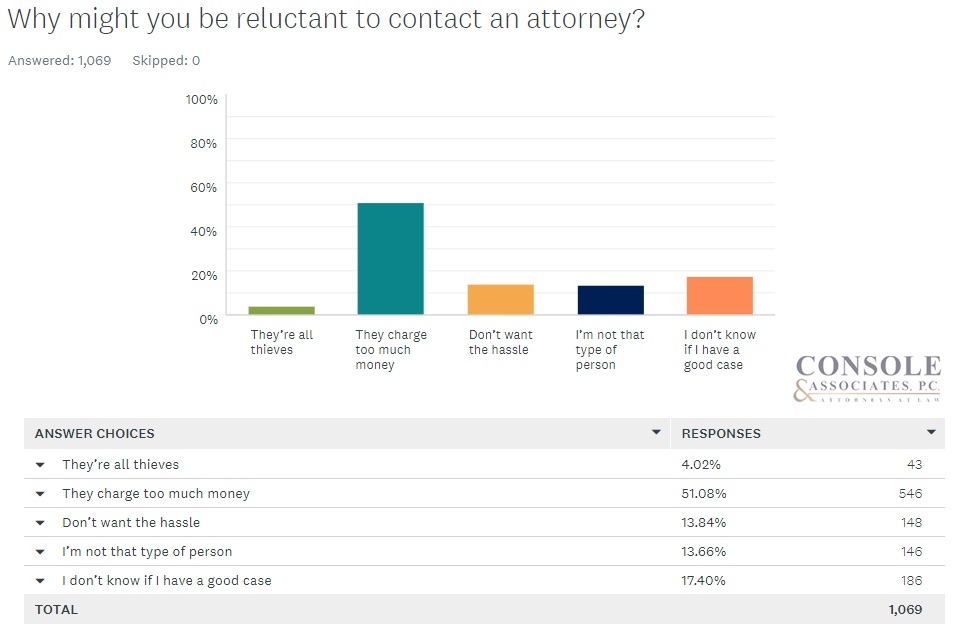
In fact, the perceived cost is the #1 most common reason why people are reluctant to hire attorneys. In an original survey we conducted of more than 1,000 people, 51 percent of the general public population bought into this misconception that lawyers charge too much money.
The truth is, medical malpractice firms in NJ don’t charge any money upfront for legal representation. Our no-win, no-fee legal services allow every patient harmed by medical errors to seek justice without taking on the financial risk of a lawsuit.
At Console & Associates, the consultation is free, and you’ll never have to pay anything upfront for our representation.
2. Fear of not understanding the legal process
You might be concerned that you don’t know what to expect from a medical malpractice lawsuit. Relax! Our medical malpractice lawyers are here to help, not to cause you any more stress.
Moving forward with a medical malpractice claim starts simply enough. We’ll gather as much information as we can about you and your situation. Then we will conduct an initial investigation into your medical treatment to determine if you have a case.
We will explain the legal process and what you need for a medical malpractice case. Once you understand the process, a lawsuit won’t seem anywhere near as nerve-racking. And the best part is that your attorney will handle every aspect of your legal process for you – so there’s no hassle and no distraction from working toward recovery.
3. Fear of not knowing where to start
It might be hard for you to clearly and easily explain what happened to you. After all, you’re not a doctor – but we don’t expect you to be.
Just tell us what you know and answer our questions as best you can. Once we’re familiar with your situation and whether or not you might have a case, we can review your medical records and investigate the details that even you might not be sure of.
4. Fear of having to give up control
When you’ve suffered any kind of accident or injury, it can feel like your life is spinning out of your control. Putting your trust in an attorney may feel like giving up even more of your control, but it can also free up your time and energy. With a lawyer on your side, you can get the financial compensation that can help you regain control of your life.
You want to know that whoever you trust with your claim has the same goals for your case as you do. With our No Win No Fee Promise, our goals are perfectly in line with yours. We want to get you the most money possible for your situation.
We make more money when you get more money, so our interests will always line up with yours. And if you don’t have a claim, we won’t try to convince you otherwise. We don’t want to waste your time (or ours) if we know you don’t have a case.
Medical malpractice is a particularly complex area of law. There’s a lot to know about medical malpractice negligence claims, including what malpractice means and how to prove it.
Medical malpractice is defined as any negligent action on the part of a healthcare professional that deviates from or falls below the standard of care and results in harm to a patient.
Deviating from the standard of care may mean doing something that the average doctor wouldn’t do, like prescribing dangerously addictive medications for an inappropriate purpose or length of time.
It can also encompass omissions, or failures to act – things like neglecting to order a test that a reasonable doctor would have done based on the patient’s symptoms and health history.
The standard of care, in general, and as it pertains to their field of medicine specifically, guides a doctor’s medical decisions and informs the choice and timing of diagnostic tests and treatments.
Falling below the standard of care can cause serious harm to a patient, including causing long-term health problems. Sometimes these errors can cost a patient their life.
Another term you may hear is medical negligence. Medical malpractice and medical negligence are often used simultaneously because these two elements of a claim are closely related – so much so that patients may not be able to differentiate between them. For experienced attorneys, the small but important difference between medical malpractice vs. medical negligence comes down to whether there was intent, such as a conscious decision to do (or not do) something even though it could raise the risk of harm to the patient.
Once an attorney is familiar with the details of your claim, they can help you understand whether your case fits better under the banner of medical malpractice or medical negligence. In either situation, you may be able to pursue a case. The legal points your attorney makes in that claim that is likely to be different depending on whether they are claiming malpractice or negligence.
Defining malpractice without talking about the standard of care is virtually impossible. That’s because the negligence that occurs in a malpractice situation hinges on whether or not the healthcare provider deviated from the standards established by the broader medical community.

Bad Bedside Manner Or Medical Malpractice
It’s important to understand the difference between a negative outcome and poor quality of care. If you have only one of these elements – but not both – then you may not have the grounds for a claim.
When medical malpractice occurs, negligence in some form has led to a medical mistake or error on the part of the healthcare provider. If, however, that error doesn’t bring about a bad medical result, you have no claim.
This could happen because the mistake was detected quickly by the person who made it or by someone else, or by sheer luck that the worst consequences didn’t occur. Since you didn’t suffer further injuries or complications that resulted in considerable physical or financial losses, you won’t be able to pursue a claim in this situation.
Another time when you have no claim is if you have a bad medical outcome, but no malpractice occurred. Unfortunately, bad medical outcomes happen all the time, and there might not be anyone to blame.
Understanding why your surgery was not successful, and what consequence the failed surgery had on your health, is a large part of determining if you have a claim.
It may be that your surgery did not fix your problem, at all or to the extent you were hoping it would. This might not be because your doctor did anything wrong, but because your condition was too advanced for modern medical interventions to fix or because your body simply didn’t respond well to the treatment. If the surgery didn’t pose further problems for you, then it’s likely that the failure to fix your existing problems won’t be considered harm enough to warrant pursuing a claim.
What if you did suffer an injury? Every surgery has risks, even when performed by a highly qualified and very careful doctor. You could be harmed as a result of suffering one of those predictable risks, but you still wouldn’t have a claim. On the other hand, you could pursue a claim if what happened to you was not a known risk of surgery but instead the result of a botched technique, an impaired or overconfident doctor, or another negligent action.
A wrong diagnosis may be more likely than an unsuccessful surgery to bring about a medical negligence claim. Remember, malpractice refers to situations in which a healthcare professional negligently or carelessly strays from the standard of care. The question becomes whether the wrong diagnosis or delayed diagnosis happened because of negligence or merely because you matter was a medically difficult case.
Suppose your condition is exceptionally rare and presented very few of the symptoms associated with it. Your doctor’s inability to diagnose that condition correctly may be understandable, especially if you don’t have the most common risk factors for that condition and your doctor is not a specialist in that type of disease. It’s possible that your doctor did all the right things that the average member of the larger medical community would have done but still failed to make that correct diagnosis in a timely manner.
On the other hand, it’s a totally different story if your doctor overlooked key symptoms or brushed past significant risk factors in your medical history. If your doctor neglected to order the diagnostic testing that a reasonable doctor would’ve done, or if that physician failed to interpret your test results accurately, then you might have a claim for malpractice based on a wrong diagnosis.
At this point, you need to successfully show that the wrong or delayed diagnosis caused you additional harm. Did the delay, for example, allow your condition to worsen? Did you develop serious side effects or complications because of the unnecessary treatments you underwent for a medical condition you don’t have?
An unwanted outcome doesn’t always mean malpractice. However, if you think that what happened to you or a loved one was preventable, you should always look into it. You have nothing whatsoever to lose by taking advantage of a free consultation with an experienced medical malpractice lawyer in NJ.
If you think you have been the victim of a medical error, you might move forward with a medical malpractice claim. A medical malpractice claim is a type of insurance claim in which you would seek financial compensation from the health professional’s malpractice insurance carrier. Think of this as a policy that provides benefits to an injured party.
Your claim turns into a medical malpractice lawsuit when you commence litigation. This means you have filed legal documents known as a complaint, or pleading, with a court of law.
Filing a medical malpractice lawsuit doesn’t mean that your case will go to court. Most medical malpractice cases don’t. What it means is that you have the option to take the matter to trial if that’s what’s necessary to get the money you deserve for your claim.
You can have an attorney file a medical malpractice lawsuit on your behalf and still settle the matter out of court through negotiations with the malpractice insurance company. In fact, that’s often the goal – to reach a settlement that covers the full extent of your injuries and losses before your claim progresses to the courtroom.
If you’re wondering about the qualities of medical malpractice vs. personal injury, you’re right in thinking that these two types of law are connected. Medical malpractice is one type of personal injury matter.
Generally, personal injury encompasses any civil legal matter that revolves around injuries sustained to your physical person as a result of negligence.
There are, however, a few important differences between medical malpractice and personal injury claims. In a medical malpractice claim:
Like a personal injury, medical malpractice is a type of tort claim. Tort cases are civil legal matters that revolve around a person suffering some type of harm or loss because of another’s actions, whether those actions were negligent or intentional.
A civil matter simply means that the defendant is not facing criminal charges but rather consequences for non-criminal acts of wrongdoing. Your case will bring about a financial payout, as opposed to jail time for the defendant. You don’t necessarily have to prove that a law was broken, just that the person – in this case, a medical professional – failed to act with reasonable care to uphold a duty they owed to you.
That said, it’s possible for the defendant to face criminal charges related to your injury in a separate legal matter.

A medical malpractice lawyer is an attorney who routinely handles medical malpractice claims. In fact, lawyers who handle medical malpractice claims sometimes focus their legal practice exclusively on medical negligence and other personal injury matters.
Medical malpractice lawyers are qualified to practice any area of law. They go to law school and pass the Bar Exam, just like general practice lawyers do. The difference between general practice lawyers and medical malpractice lawyers isn’t one of the qualifications to practice law, but instead one of experience.
Successfully resolving a New Jersey medical malpractice claim takes more than you learn in law school. It requires developing a deeper level of familiarity with this branch of law and with the strategies and practices that achieve a favorable resolution. Winning your medical malpractice matter means understanding medical negligence – negligent actions unique to the practice of healthcare providers – and how to present compelling arguments on behalf of the patient.
You gain these proficiencies through experience. Due to the complexity of this area of law and the expense of pursuing medical negligence claims in New Jersey, attorneys who have a track record of achieving results for their clients may decide to handle med mal and personal injury matters exclusively.
Medical errors take many forms. Over decades of handling misdiagnosis and surgical malpractice cases, over-prescription claims, medical device error claims, and other matters of medical negligence, our trial attorneys have developed a breadth and depth of experience handling matters like yours. We can draw on this experience to get you the results you deserve.
Does your situation fall under medical malpractice? That depends on a lot of factors and details that an attorney would need to examine carefully. Every medical negligence claim is unique, with different negligent parties, deviations from the standard of care, and outcomes.
Examples of medical malpractice can include:
Diagnostic failures are one of the most common types of claims involving medical malpractice. After all, if your medical condition isn’t diagnosed correctly, how can it possibly be treated correctly?
Experienced attorneys who handle medical malpractice know the differences between distinct types of failure to diagnose cases, such as:
Incorrectly diagnosing you with a different medical condition than the one you actually have. Misdiagnosis is particularly dangerous because your actual medical condition isn’t being addressed and you may be receiving unnecessary and harmful treatments for a condition you never had in the first place.
A prompt and accurate diagnosis is the first step to treating any medical issue. When the diagnosis is delayed or just plain wrong, the condition may progress to something more serious that is harder to treat effectively.
Doctors and other healthcare providers may make a few different kinds of erroneous diagnoses. A misdiagnosis occurs when a doctor arrives at the wrong diagnosis and, potentially, begins treatment for a condition you don’t actually have. In a missed diagnosis, a physician may dismiss your symptoms entirely and neglect to treat them. A delayed diagnosis occurs when, instead of diagnosing and treating your issue promptly, doctors delay reaching a diagnosis or formulating a treatment plan.
In a missed diagnosis, your doctor fails to assign any diagnosis to your symptoms or complaints. Perhaps the doctor brushes off your complaints as “normal” consequences of getting older, pregnancy, or physical labor or activity, without offering any real treatment option. Your doctor might also dismiss valid health concerns as “anxiety,” often without addressing the problem. In some instances, there’s an overlap between misdiagnoses and missed diagnoses.
In the case of a delayed diagnosis, your actual medical condition is missed or misdiagnosed initially. By the time you receive the right diagnosis – whether because you sought a second opinion or because you continued asking the dismissive doctor about it until the right tests were finally performed – your condition was worse.
The delay in finding out your medical condition allowed further damage to occur or the disease to progress to a more advanced stage. Delayed diagnosis cases usually involve a worsened prognosis. The patient may need to undergo more invasive and expensive medical treatments than would likely be necessary if diagnosed early.
In some particularly tragic circumstances, the delay has allowed the illness to progress to a terminal stage that modern medicine is not advanced enough to treat effectively.

Diagnosing a patient’s medical condition accurately isn’t always easy. However, doctors undergo lengthy education and training to become proficient in this crucial part of the practice of medicine.
Often, doctors who are facing allegations of medical malpractice for misdiagnosis have failed – in one or more ways – to uphold the standard of care set by the medical community.
Some of the most common causes of misdiagnosis and other diagnostic errors are:
A failure to diagnose attorney can determine what your provider could or should have done differently. It costs you nothing to have us examine your full medical history and the records of your interactions with the negligent doctor and to consult a medical expert in a relevant field about the unique circumstances of your claim.
A particularly devastating area of medical malpractice misdiagnosis claims is medical negligence. For most types of cancer, early detection and treatment are key to surviving or having the best possible prognosis.
That’s the reason why cancer survival rates are distinguished by stage. Treatments for early-stage, localized tumors are far more effective than those attempting to treat metastasized cancers that have spread to distant regions of the body. Any significant delay in getting an accurate diagnosis and commencing treatment may allow cancers to grow and spread, reducing the chance of a positive outcome.
Some of the types of cancers that commonly go misdiagnosed include:
If you were diagnosed with any form of late-stage cancer and wonder if your doctor should have caught it earlier, you need to consult a failure to diagnose cancer attorney.
We can delve deep into your medical chart and history and, with the help of medical experts in the field, determine whether your doctor strayed from the standard of care by brushing off a telling symptom or neglecting to order the test that would have resulted in early detection.
A failure to diagnose cancer claim could remove any financial barriers to accessing the best treatment and ease the financial burden your family is facing.
You may also have a medical claim if you were misdiagnosed with a cancer you didn’t have, if you suffered measurable harm due to this diagnostic error. Undergoing radiation, chemotherapy, invasive surgeries, and other cancer treatments unnecessarily can raise your risk of developing cancer in the future and cause you very real physical, emotional, and financial harm. An unnecessary treatment attorney may be able to help you understand your legal rights and options.
Cancers are far from the only types of medical conditions that go undiagnosed. The failure to diagnose any illness could potentially be the grounds for a medical malpractice claim if the delay in getting the right treatment harmed your health in a significant way.
Our NJ misdiagnosis/delayed diagnosis attorneys are familiar with the challenges of claims involving the following types of diagnostic errors:
If you believe you were the victim of a missed or delayed diagnosis of any serious medical condition, the time to take action is now.
Diagnosing a medical condition correctly is important, but an accurate diagnosis alone isn’t enough to make things better. Doctors who make errors treating their patients’ medical conditions – which can happen in any number of ways – can do more harm than good.
Incorrect treatment of a medical condition may include:
If their treatment mistakes cause additional problems or complications, healthcare professionals may face consequences in the form of a medical malpractice lawsuit.
One particularly alarming form of treatment error is surgical errors. Surgeries, operations that allow physicians to intervene in and manipulate the inside of the body, can leave patients vulnerable to substantial harm if their providers are not cautious.
Healthcare and medical professionals may commit surgical errors in many different ways, including:
Not all matters of surgical malpractice occur while you’re on the operating table. In many ways, postoperative negligence can be just as devastating as a slip-up during surgery.
Postoperative negligence encompasses any deviation from the standard of care that occurs during the period following surgery, in which the patient should be monitored. These scenarios can include:
A lot can go wrong when a patient’s healthcare team fails them during the postoperative period. Some of the complications our New Jersey medical malpractice lawyers have encountered due to postoperative surgical negligence include:
A variety of infections of various types may occur following surgery, including:
Infections aren’t always a clear sign of surgical error negligence, but they can indicate unsanitary conditions or, at times, inadequate monitoring of the patient. If you feel that your infection may have resulted from a healthcare professional’s negligence or that it wasn’t caught and addressed in a timely manner, it’s worth seeking a medical malpractice free consultation with an attorney.
It’s also possible for patients to pursue a claim for preoperative negligence if their medical providers did not adequately prepare them for surgery. For example, if your doctors, nurses, and other members of your healthcare team failed to take a pre-operative assessment and physical exam, this could be a factor in negative outcomes like administering too much or too little anesthesia.
Surgical errors can occur in hospitals or in ambulatory surgical centers. Both inpatient and outpatient surgeries may allow opportunities for malpractice to happen.
Just a few examples of instances in which you might need to contact a surgical errors attorney include:
Pharmaceutical medications are widely used in the practice of medicine, but virtually all drugs carry some risks and potential side effects. A healthcare professional who is careless in prescribing or administering medications could pose a serious threat to their patient, causing long-term and catastrophic injuries, addiction, or deadly drug interactions or overdoses.
Medication error malpractice encompasses deviations from the standard of care.
A prescription error doesn’t always cause the level of harm that rises to the grounds for a med mal case. When medication errors do result in a malpractice lawsuit, it’s usually because the mistake led to severe harm.
Too much of any medication has the potential for harm, but for powerful painkillers called opioids, that danger is particularly pronounced. Opioids are medications that belong to the same class of drugs as the illegal drug heroin and affect the body in much the same way as this illicit substance.
Opioids have a known potential for addiction and dependence and may be fatal if an overdose occurs. A doctor may contribute to these risks through negligent behaviors like the following:
If you believe you or a loved one may have been over-prescribed opioids by a doctor, it’s important to get medical and legal help right away. An opioid-related medical malpractice attorney may be able to help if addiction or overdose has occurred. If you realize the danger in time to prevent a tragedy, you may be able to get help exploring your options for getting appropriate treatment for your loved one before it’s too late, as well as holding a doctor accountable.
The medications used to induce numbness and loss of consciousness during a medical procedure are known as anesthetics or anesthesia.
These drugs are crucial to allowing doctors to perform procedures and keeping patients comfortable during what would otherwise be a painful medical intervention. Used incorrectly or without appropriate caution, though, anesthesia can be very dangerous.
Too little anesthesia could leave a patient in the nightmarish situation of feeling everything during surgery but being unable to communicate, a traumatic experience that can understandably cause patients to develop symptoms of post-traumatic stress disorder. Too much anesthesia could, at an extreme level, lead to oxygen deprivation and brain damage, which may be fatal. Additionally, giving a patient the wrong medication—such as an anesthetic agent that they are allergic to—can also cause them to suffer serious harm.
Bad responses to anesthesia don’t always constitute malpractice. However, serious negative outcomes from anesthesia are rare when providers adhere to the standard of care, taking steps such as carefully examining the patient, taking their complete medical history, and monitoring their vital signs during the procedure and postoperative recovery.
Some of the negligent actions that may lead to anesthesia accidents include:
Any of these serious mistakes could put the patient in danger. An overdose of anesthesia can lead to severe brain damage or death. If too little anesthesia is used, the painful experience of being awake during surgery can be traumatic enough to lead to debilitating mental health conditions like post-traumatic stress disorder (PTSD).
The stress of anesthesia itself can also lead to strokes, heart attacks, and other serious medical events, particularly in patients with certain risk factors. That’s one important reason why the healthcare practitioners attending to an anesthetized patient need to be fully aware of the patient’s health history, allergies, pre-surgical testing results, and other risk factors.
Specialized doctors called anesthesiologists and Certified Registered Nurse Anesthetists (CRNAs) are the healthcare professionals most qualified to administer anesthesia. Your care team may also include an anesthesia assistant. Physicians who do not specialize in anesthesiology may also administer anesthetics.
If any of these care providers or other healthcare practitioners on your surgical team contribute to an anesthesia accident, you may have the right to sue that professional for the harm they’ve caused you. The hospital or outpatient surgery center may also be liable for the medical error. An anesthesia malpractice attorney can identify all possible defendants in your medical negligence claim and sort out precisely how each party contributed to the accident.
Birth-related injuries are injuries suffered during the birth of a child.
Pregnancy, delivery, and the postpartum and neonatal periods can be fraught with complications and risks for both mother and baby. When a doctor fails to detect and take appropriate action for these complications or acts negligently during delivery, it can result in birth injuries that change the course of the newborn’s life, as well as the future of their family.
Some examples of medical malpractice in birth injuries include:
Obstetrical malpractice, like these and other forms of medical negligence, can have serious consequences. A newborn who suffers brachial plexus injuries – such as Erb’s palsy – due to medical malpractice committed during delivery may need months of physical therapy. Some infants may even have to undergo surgery. If the medical error led to oxygen deprivation and an anoxic brain injury, the baby could have severe, lifelong physical and cognitive deficiencies.
In the case of some serious birth defects, parents may have the right to pursue a wrongful birth case against a medical practitioner for failure to diagnose genetic conditions. Families sometimes pursue a case of this nature to seek compensation for the lifelong, round-the-clock care the child will require as well as the pain and suffering the child – and their family – has been forced to undergo as a result of the doctor’s deviation from the standard of care.
When a medical mistake strips away the joy that should have accompanied the birth of a child, it’s time to consult with a birth injury attorney. Your lawyer will be an advocate for your child, helping your family get the compensation and access to care that can be instrumental in reaching the best possible medical improvement and quality of life.
Cerebral palsy, the most common childhood motor disability, isn’t always caused by medical malpractice. When it is, however, the consequences can be devastating.
Just over half of children with cerebral palsy will ever walk independently, and nearly one-third of children with this condition have limited or no walking ability, according to the Centers for Disease Control and Prevention (CDC). Parents often worry about the future of their children with cerebral palsy, and they have good reason to be apprehensive. The lifetime cost of care for a person with cerebral palsy is nearly $1 million, the CDC reported.
Emergencies occur every day in the United States, with 139 million ER visits reported during 2017, according to the CDC. Mistakes made in these high-stakes circumstances, when patients may well be facing life-or-death medical events, can have particularly dire consequences.
Emergency room delays and mistakes can include failures like the following:
Although emergency departments can be hectic and fast-paced environments, there’s no room or excuse for negligence in these settings. Emergency room errors that result from a healthcare professional deviating from the standard of care may lay the grounds for a med mal lawsuit.
Despite the challenges, the physicians and other healthcare professionals who work in an Emergency Room must adhere to the standard of care. That means they can’t ignore complaints of symptoms that could indicate a serious problem or allow patients’ conditions to deteriorate while they wait to be seen. A misdiagnosis or delayed diagnosis that would not have happened had the ER physicians and other providers adhered to the standard of care may also prompt a medical malpractice lawsuit.
Dealing with emergency room bills and the costs of follow-up appointments is enough to worry about financially as it is. You can get no-win, no-fee assistance from experienced emergency room malpractice attorneys to better understand your options and legal rights.
Many instances of medical malpractice take place in hospitals. These medical facilities hold massive numbers of patients. Their providers often work long shifts, including overnight hours, which can contribute to drowsiness. Patients often come to the hospital for treatment for severe conditions as well as for surgical procedures, which makes them more vulnerable to types of malpractice like surgical errors, anesthesia errors, and medication errors.
It’s often difficult for a patient to know if malpractice occurred at a hospital or figure out how to hold the hospital and its staff accountable. Fortunately, a hospital negligence attorney can handle the monumental task of identifying all of the providers and medical facilities that acted negligently and how they strayed from the standard of care. It costs you nothing to speak to an attorney for medical malpractice hospital errors about your legal rights and options.
All medical facilities, as well as individual practitioners, are responsible for providing patient care that meets the standard of care. You may be able to move forward with a lawsuit against a variety of types of hospitals and medical facilities, including:
Suing a hospital doesn’t negate your doctor’s accountability for malpractice. However, it allows you to hold responsible for your injuries both the individual healthcare professional (or more than one) and the health system that contributed to and enabled their negligence.
Nursing home abuse and neglect is a significant problem in the United States. Not all instances of elder abuse and neglect in nursing homes constitute medical negligence. However, nursing home negligence can be considered malpractice in situations like the following:
A nursing home abuse attorney can determine if neglect occurred, if it rises to the level of malpractice, and what options you have for protecting your family member from further harm and for securing compensation.
When you think of medical malpractice, you probably imagine a claim against a doctor you’ve seen face-to-face, often over some length of time. Medical negligence cases involving laboratory errors may mean suing a provider you haven’t even met – but whose job was crucial to diagnosing your medical conditions or checking for changes in your health. Laboratory errors include:
Suing a facility or provider for laboratory errors and mistakes requires evidence not only of the laboratory’s negligence but also of the real, measurable harm you suffered as a result.

Defective medical products commonly lead to claims against the manufacturers of those devices. However, there are times when you may have the grounds for a claim against your doctor or another healthcare professional. These instances include:
The reason medical devices fail to work correctly isn’t always dangerously defective design or manufacturing. Sometimes the medical device failures that leave patients seriously injured result from user error on the part of the healthcare worker. The more complex and necessary the medical equipment is—such as a ventilator that sustains the life of a patient who can’t breathe on their own—the greater the risk of a device error causing serious harm. Medical negligence lawsuits that involve mishandling medical equipment and devices may involve other parties besides physicians, including home health aides, nursing assistants, and nurses.
This isn’t an exhaustive list of all of the ways healthcare providers can deviate from the standard of care to the detriment of their patients. If your situation doesn’t fit into one of these categories but you still suspect that medical negligence may have occurred, it’s time to speak to a medical malpractice law firm directly (at no charge) about your claim.
A mistake with a medical device can have catastrophic consequences. For example, if a home health aide negligently handles ventilator equipment, a patient who depends on that machine could go without oxygen long enough to sustain a life-threatening anoxic brain injury.
A medical equipment failure attorney can help you get to the bottom of what happened and who is really to blame for a serious injury involving a medical device.
You may have noticed that a lot of instances of medical negligence involve what doctors fail to do, rather than what they do. These failures may occur in any type of healthcare setting or medical specialty. Sometimes referred to as negligent omissions, the types of failures that can constitute medical negligence may include:
Any omission that constitutes a deviation from the standard of care could potentially be the grounds for a medical malpractice claim – from allowing a patient to fall out of bed to the failure of staff at a psychiatric hospital to prevent suicide.
If you believe some form of negligent omission played a part in your or your loved one’s negative health outcome, medical malpractice and hospital negligence lawyer can unravel the complicated strands of liability.
Healthcare professionals can also face consequences when they stray from the standard of care by doing too much. The proverb “better safe than sorry” doesn’t apply when the unnecessary treatment itself causes substantial harm to a patient.
Some of the ways unnecessary treatment can amount to medical malpractice include:
According to ProPublica.org, more than 600,000 patients underwent unnecessary medical treatments in just one year.
You don’t need an unnecessary surgery attorney if your doctor ordered reasonable testing or treatments that may not have been strictly necessary but didn’t cause any harm. It’s when these unnecessary treatments have put you through considerable pain and suffering, caused you physical damage in your range of motion or sensation, made you vulnerable to new health risks, or otherwise impacted your health in a serious way that true malpractice occurred.
As a patient, you have rights. Your doctor isn’t automatically above question or reproach merely because they have a medical license. In certain instances, it’s possible for a patient to sue their doctor over issues of consent.
In ordinary situations, your doctor should get informed consent from you prior to administering a test or procedure. The best doctors communicate clearly and honestly with their patients. They cover the benefits and risks and work with the patient to make choices that are reasonable for the unique situation. They respect their patients’ decisions and concerns.
The problem is that other doctors don’t respect patients’ wishes or their consent. Situations in which consent issues are considered a form of medical malpractice include:
There are certain instances, such as medical emergencies, when getting the consent of a patient – or even their family – isn’t possible. Doctors may need to take action immediately to save a patients’ life, limb, or function.
However, there’s a massive difference between situations so urgent that doctors can’t waste precious minutes trying to get a patient’s consent and the arrogant physicians that routinely stomp on their patients’ rights and disregard rules of consent.
Malpractice lawsuits over consent in medical procedures often arise when a complication or risk that a patient should have been informed of – but, because of the doctor’s negligence, was not – occurs and poses serious consequences. An attorney may be able to prove that your doctor deviated from the standard of care by failing to get full medical negligence from you prior to the procedure.
Medical negligence can occur in any specialty of medicine, even ones you wouldn’t necessarily think of. Some of the specific types of medical specialties in which a doctor or other healthcare provider may commit malpractice include:
This is far from an exhaustive list of specialties in which malpractice can occur. New Jersey’s malpractice attorneys at Console & Associates are prepared to handle medical error lawsuits of all varieties. We can help you start getting answers with a free consultation.
Not all medical malpractice matters fit neatly into a single category. Some instances include negligent actions that encompass different types of mistakes and omissions. There may have been one key deviation from the standard of care or a sequence of errors that, together, allowed the negative health outcome to occur.
For example, our compartment syndrome attorneys have seen malpractice – usually in the form of a failure to diagnose this painful condition – occur in different kinds of circumstances. A case might constitute emergency room negligence if the patient comes to the ER with an injury, or it might fit into surgical malpractice if the patient developed compartment syndrome when undergoing planned orthopedic surgery. The same is true for other conditions with multiple causes, such as Complex Regional Pain Syndrome.
Similarly, a hospital malpractice claim often involves other specific types of medical negligence, like misdiagnosis, surgical error, or emergency room mistakes.
You don’t have to know precisely who was negligent, or how, when you reach out to a medical malpractice lawyer. Figuring out all relevant theories of liability and how to prove negligence on the part of every defendant is among the legal services we provide for our clients. We’ll look at the details of your unique situation from every angle to identify all possible instances of medical negligence.

Medical errors can lead to all kinds of complications and injuries, large and small. But the types of mistakes that most commonly lead to malpractice claims are the ones that have irreversible consequences.
Often, this means a catastrophic injury, one that will have a serious and long-term or permanent impact on the patient’s life.
Injuries that lead to paralysis are some of the most serious injuries you could sustain. Paralysis refers to the loss of the ability to move and, in some cases, the loss of sensation.
Paralysis may be temporary or permanent. It can be partial or complete. It may occur on only one side of your body or on both sides, as well as in different regions of the body.
Types of paralysis include:
It’s hard to overstate the impact paralysis can have on your life. Even the smallest, most routine tasks of daily life require you to have the mobility that you lose when paralyzed. A medical error that leaves you paralyzed can steal your independence from you. Without assistance – in the form of caretakers and medical devices like wheelchairs – you can’t get around or take care of yourself.
The financial impact of paralysis can be as catastrophic as the physical impact. In the first year following your injury, you can expect to have medical expenses between $375,196 and $1,149,629, the National Spinal Cord Injury Statistical Center reported.
Every subsequent year will pose tens, if not hundreds, of thousands of dollars in medical expenses. For the least severe level of motor function, medical expenses for one year are, on average, $45,572, while medical costs for the most severe form of tetraplegia average nearly $200,000 per year.
Someone has to bear these unbearable costs, and it shouldn’t be your family – especially when your injury was someone else’s fault.
Medical malpractice can lead to paralysis in a variety of ways, including:
Your healthcare providers may not admit to you that malpractice is what caused you to become paralyzed. But, when paralysis isn’t the result of an accident or similar injury but instead occurred after a medical procedure or as a result of a medical condition that you suspect was misdiagnosed, you may have a claim.
A paralysis attorney can help you understand your legal rights and options and draw from our knowledge of working with paralyzed claimants to make sure your claim for compensation covers all of your current and future expenses.
Brain Injuries can have a devastating impact on physical and cognitive function. Much like injuries that cause paralysis, brain injuries can impact your mobility and your independence. They can also affect your memory, your ability to communicate, and even your personality.
Medical malpractice can lead to both anoxic brain injuries – damage that occurs to the brain due to a lack of oxygen – and traumatic brain injuries.
Some of the different types of malpractice that can lead to a brain injury include:
When a healthcare professional’s mistake leads to a brain injury, the results can be debilitating, if not deadly.
Survivors of a severe traumatic or anoxic brain injury may require round-the-clock care. Even when a brain injury is relatively mild, it may have a significant impact on your life. Brain injury survivors may need medications, surgery, and physical, occupational, cognitive, and speech therapy to reach their maximum level of improvement. Even then, they may still experience some deficiencies in functioning.
Some brain injury survivors are ultimately able to care for themselves independently, but they may need the use of some assistive technologies, medical devices, and cognitive strategies to adjust to the new normal that is life after a brain injury. For other brain injury survivors, life can feel like a shell of what it once was, and they may need help with even the most basic of tasks.
If you believe that medical negligence is the reason you or your loved one suffered a brain injury, a free case evaluation can help you get answers and get the ball rolling on your malpractice claim. A brain injury isn’t the kind of harm you can just “bounce back” from, and it’s crucial that the practitioners who caused this injury to face the consequences.
In some particularly drastic instances of malpractice, a patient may lose a limb due to the healthcare professional’s mistakes.
This kind of loss can change your life forever. You deserve a medical malpractice payout that fully compensates you for all of your struggles as an amputee.
Limb loss that arises from malpractice is often due to a medical error that leads to a preventable amputation. This situation can occur in many different ways, including:
Medical errors that result in limb loss can be as dramatic as waking from surgery to the horror of learning that your healthy limb has been amputated. They can also be far more subtle and difficult to detect.
Simply put, if you were surprised by the need for amputation and wonder if your doctor could have done more to prevent the loss of your limb, you need to speak to an amputation medical malpractice attorney. That’s the only way you will get answers to your questions about what happened and what to do next.
Catastrophic injuries can also refer to:
These types of catastrophic injuries are among the harms our New Jersey medical malpractice lawyers encounter most often, but this isn’t an exhaustive list of the injuries for which you could seek compensation.
Anytime you have an injury that you suspect resulted from malpractice and that has had a significant negative influence on your life – physically, emotionally, or financially – you owe it to yourself to find out your legal options.
Taking advantage of a free case evaluation can help you understand what happened and what to do next. The worst that can happen is that you find out (at no cost) that your claim would cost more to pursue than you would be able to get for it based on your damages.

Instances of medical malpractice resulting in death are, unfortunately, all too common in the United States. If you believe that a doctor or another health professional’s negligence was what cost your loved one their life, there’s a strong possibility that your suspicions could be correct.
Families who have lost a loved one to a medical error may have the right to hold the provider accountable through a medical malpractice or wrongful death lawsuit.
When suing for medical malpractice deaths, you can finally get answers to the questions you have been wondering about – like what really happened to cause your loved one’s death and why this occurred.
These answers, and ensuring that the negligent medical professional faces accountability for their actions, can often help families find some closure. The compensation your family receives can help you better cope with the financial hardships that may arise in the aftermath of a medical malpractice death.
Medical malpractice claims are complex and fact-specific. To determine if medical negligence occurred, an attorney would need to review all available information. In the case of a patient who passed away due to a negative health outcome or complications, this should include a review of a thorough autopsy performed by a qualified medical examiner.
In some instances, a family may not suspect malpractice until after some time has passed. This is completely understandable. You’re in shock over the loss of your loved one, and you trusted their doctor. But it means that, by the time you start to think about a medical malpractice claim, it may be too late to have an autopsy performed.
Doctors may have told you that doing an autopsy is unnecessary or tried to deter you from proceeding with one. Whatever the case, you may be wondering now if not having an autopsy means you can’t move forward with a claim.
The answer is, honestly, that it depends on the situation. If there is enough evidence available from other sources – like your loved one’s medical records – to prove that malpractice was what led to the untimely death, you may be able to sue even without an autopsy. In other instances, no matter how compelling your theory of what occurred maybe, there’s no proof without an autopsy report.
Always be upfront when consulting an attorney about a medical negligence claim, because whether or not there’s an autopsy available may affect the viability of your case. An attorney can review the available information and let you know if they feel that the evidence for your malpractice claim is strong enough to succeed even without having an autopsy.
If not, you’re no worse off than you were before consulting an attorney, since the consultation is free, and you at least have the peace of mind that you explored all of your options.
Given the heartbreaking effects of a doctor’s error, it makes sense that patients and their families often wonder why medical malpractice happens.
Doctors are highly educated in medical school and painstakingly trained in clinical practice over years of residency training. Yet medical errors are astoundingly common – so much so that as many as one-quarter of all hospital patients experience a medical error during their hospitalization, according to the New England Journal of Medicine.
Like the human body itself, the factors behind medical malpractice can be intricate and complex. Some of the reasons why medical errors occur include:
The fault for medical malpractice may fall squarely on one irresponsible practitioner who chose to go against the standard of care out of overconfidence, apathy, or for personal gain.
Or it may be a product of systemic failings, like a hospital system that saves money through consistent understaffing, regularly overworks its providers to the point of burnout, and has inadequate communication and record-keeping procedures.
Either way, the parties responsible for your injuries must face the consequences. Otherwise, your family will be the one to pay for their mistakes – and other families may face the same harm.
Research published in the New England Journal of Medicine shows that approximately 1 percent of all physicians accounted for 32 percent of paid medical malpractice claims. The same negligent doctors are ruining patients’ lives by committing medical errors over and over again. If you stay silent, the doctor has yet another chance to harm someone else.

How can medical malpractice be prevented? It’s a difficult conundrum to solve, precisely because there are so many types of malpractice and so many underlying causes of medical errors.
As a patient, the best step you can take to reduce the risk of becoming a medical malpractice victim is to advocate for yourself.
Unfortunately, these tips, while helpful, can’t prevent all instances of malpractice. In some instances, like when you’re under general anesthesia, you physically can’t advocate for yourself. And some physicians are so negligent that even a patient who advocates strongly for themselves may not be able to get through to them.
A big part of medical malpractice and prevention falls on medical facilities and healthcare systems themselves. They must take steps to:
You think you may have the grounds for a medical malpractice lawsuit. You know that a negative health outcome occurred, and you have reason to suspect a medical error was involved.
But how do you know for sure if you have a med mal claim? And how do you take the next steps to move forward with your case?
Malpractice claims arise out of medical negligence. Proving legal liability for injuries that result from what you allege was an incident of medical malpractice requires you to establish certain elements:
New Jersey medical malpractice attorneys take on cases on behalf of:
To have the grounds for a claim, you must have suffered substantial injuries due to the medical mistake. Those injuries must have had some amount of measurable impact on your life.
You can’t sue for a medical error – even if its occurrence is clearly documented – if it was immediately resolved without causing further harm. A surgeon may admit to making a mistake during an operation, but if that mistake was quickly and completely rectified in the OR without causing any additional complications, you likely have no case. You can’t sue for what could have happened, only what did happen.
Medical malpractice is negligence committed by a healthcare professional. This provider may be a generalist or a specialist.
In some cases, medical malpractice is the result of a moment of negligence committed by someone who met a patient one time in a support role for a procedure. Other times, it’s a practitioner who had a years-long relationship caring for the patient that commits a medical error. That error may be a one-time lapse of judgment or years of failing to address an ongoing issue.
For you to have a case against a healthcare provider, that professional must have had some responsibility to you as a patient. They also must have contributed in some way to the malpractice that led to a negative health outcome.
Most people associate medical malpractice with doctors and surgeons. But MDs and DOs are far from the only healthcare professionals who can be held accountable for a serious mistake.
Other providers and practitioners you may be able to sue for medical malpractice include:
Individuals aren’t the only possible defendants in a medical malpractice claim. You can also sue a facility, such as a:
Physicians of just about any specialty may face medical malpractice lawsuits if their negligence harms a patient.
Some of the types of doctors who may be sued for medical negligence include:
Medical malpractice for physicians is so much more common because doctors are the ones with the primary responsibility for a patient. However, plenty of claims involve other healthcare professionals, such as nurses and physician assistants.
Nursing is a demanding job, and a small mistake could have disastrous consequences.
Registered nurses may face malpractice claims for their roles in matters of improper or excessive medication, such as when a doctor prescribed the correct medication and dosage but the nurse failed to administer it correctly. Medical malpractice for nurses may also focus on preventable hospital infections, injection site injuries, or neglect that leaves a hospitalized patient dehydrated or malnourished.
Medical malpractice for nurse practitioners may look more similar to claims against physicians. Nurse practitioners are a type of advanced practice registered nurse. A nurse practitioner may be authorized to work independently of a doctor and to be the provider of primary or specialty medical care, prescribe treatments and medication, and order diagnostic tests. The expanded scope of work for a nurse practitioner allows for more potential for medical errors that could have serious consequences.

Physician assistants, sometimes called PAs, practice medicine under the supervision of a physician and in a more limited scope compared to doctors. Like nurse practitioners in many states, PAs are usually authorized to write prescriptions, order and interpret test results, and provide treatment for medical conditions.
Many of the clinical tasks a PA performs are similar to that of a doctor. As a result, medical errors made during these tasks can leave a physician assistant facing a medical negligence claim. Because the PA’s supervising doctor is also responsible for the patient, a medical mistake committed by a PA may result in a claim against both practitioners.
If you believe multiple healthcare professionals played a role in the negligence that harmed you, then you have the right to hold accountable every one of those practitioners.
You could file suit against any combination of individual providers and healthcare facilities that makes sense for your claim. You could also sue just one provider if you believe this individual alone was responsible for the harm you suffered.
Figuring out which parties involved in your medical care acted negligently, and in what ways, is part of your attorney’s job. Medical situations are often complicated. You may not know exactly who made a mistake.
In some cases, you may have met a provider only once, or they may have been involved in your treatment only while you were unconscious during surgery. If you saw a PA instead of their supervising doctor, or a resident who spoke with the on-call attending doctor over the phone, you may have never even met some of the possible defendants in your claim. What’s important is that your medical malpractice and negligence lawyers can establish that the provider had some duty of care to you as a patient.
Your medical malpractice law firm should meticulously review all of your medical records related to your suspected malpractice. Any party who may have been involved in the negligence that harmed you should be named as a potential defendant in your med mal lawsuit.
When is a healthcare provider liable? To have a valid New Jersey medical malpractice claim, you must establish the following elements of a medical negligence case:
When pursuing medical malpractice and negligence cases in New Jersey, your attorney must be able to show a clear, direct connection between the negligence and the damages you suffered. If a defendant can make a compelling argument that the complication you developed may have happened even if they hadn’t been negligent, that may be enough to prevent you from getting compensation.
Determining whether a provider fell short of the standard of care is challenging. That’s because, as a patient, you don’t have the medical background needed to know precisely what practices are considered in line with the standards widely considered acceptable within the medical community.
Even experienced medical malpractice lawyers in New Jersey aren’t qualified to determine, themselves, whether a deviation from the standard of care occurred. (However, after more than two decades of handling med-mal claims, we certainly can recognize which situations could potentially involve a failure to meet the standard of care.) It takes another member of that medical community that sets these standards – in other words, another doctor or healthcare professional – to provide their expert opinion on whether the defendant’s actions or omissions amounted to a deviation from these standards.
The services of medical experts are crucial in proving medical malpractice cases. When Console & Associates, NJ’s trusted team for complex medical malpractice claims, first examine a case, we identify which specialties of medicine are most relevant to your matter. We might bring in medical experts who specialize in the same branch of medicine as the potential defendant. If it’s suspected that the doctor was negligently practicing outside of their scope of practice, we might also enlist the services of a medical expert in a specialization that should have been involved in your care.
Medical experts draw on their vast knowledge of medicine, along with their professional credentials, to communicate – via written opinions or verbal testimony – precisely when, where, and how deviations from the accepted standard of care occurred based on the patient’s medical records. The findings of these medical experts are among the strongest forms of evidence you will find in a medical malpractice lawsuit.
Sometimes negligence isn’t enough. One of the things both patients and lawyers find most frustrating about medical malpractice claims is that you could meet all of these medical malpractice guidelines, but it doesn’t mean you can sue.
Even when a doctor has been negligent and there’s enough proof of that negligence that expert witnesses would agree, there isn’t always a case. Medical malpractice without injury – or at least, without significant, permanent injury – may be the grounds for an official complaint filed with state regulators, but it may not be sufficient to sue for.
Besides medical negligence, a successful medical malpractice claim hinges on having damages that are clearly caused by the mistake and are clearly significant. If your injuries weren’t permanent, if they didn’t change your life forever, then you might not be able to bring a claim – even if they put your life at risk.
See, the law doesn’t allow you to sue for what could have happened, only what did happen – a fact that understandably seems unfair to victims who underwent pain, suffering, stress, and fear. Almost dying, spending days in intensive care, needing additional surgery – most of us think these consequences are serious, but without additional damages, they aren’t always enough to warrant a medical malpractice claim.
Sometimes doctors fail to meet the standard of care and their patients suffer because of it, but there’s still no way for these patients to get compensation because their damages aren’t significant enough to outweigh the tremendous cost and burden of bringing a claim.
That’s why, apart from negligence, the most important element that must be proven in a medical malpractice case is damages.
When you work with a skilled New Jersey medical malpractice attorney team, you’re going to hear the word damages. This is the legal term for the harms and losses you suffer, in many different forms, because of your provider’s negligence.
A different but related term, money damages, refers to the compensation you recover from your claim. The amount of money damages you get is based largely on the extent of your damages. The reason why money damages are awarded is to compensate you for all of the harms and losses you have suffered.
Some of the types of damages which medical malpractice lawsuit settlements should cover include:
The average settlement for medical malpractice claims is high—$329 565, according to the Journal of the American Medical Association (JAMA). Some of the reasons medical malpractice claims tend to have such a high value are because the need for medical expert opinions to prove a deviation from the standard of care makes these claims expensive to pursue and because injuries must be serious to move forward with this type of personal injury claim.
Generally, the damages you pursue in a medical malpractice claim are considered compensatory damages. They’re intended to compensate you for the different kinds of harm you suffered. The two main kinds of compensatory damages are:
Another type of damage that sometimes comes into play in the case of a medical negligence claim is punitive damages.
The economic damages you suffer due to a medical error have definite dollar amounts associated with them. You can calculate or project the financial cost of these losses in a way that you can’t with non-economic losses.
Some examples of economic damages include:
Your non-economic damages are priceless, in a literal sense. Unlike medical bills or lost wages, you can’t calculate precise figures for your non-economic damages based on hard numbers like the amount billed for a procedure or salary expectations for the remainder of your work-life.
Some examples of non-economic damages include:
One key difference between economic and non-economic damages is that no amount of money can ever truly replace your non-economic damages. The pain and suffering, the emotional distress, the disfigurement – none of these things will be fully erased by a medical malpractice payout.
That said, getting compensated for these very real if subjective, losses are important for malpractice victims and their families to move forward. The money you recover for your non-economic losses can help you make changes to your life that help address these losses. You can use your medical malpractice payout to:
To get the most money for your claim, you need a medical malpractice lawyer with the experience to fully understand all of the ways medical negligence has impacted your life. Our New Jersey attorneys will fight to get you every dollar you deserve.

In certain instances, your medical negligence lawsuit may bring about punitive damages. Punitive damages are awarded in a trial by a judge or jury to punish the defendant.
Under a statute known as N.J.S.A. 2A:15-5.12, punitive damages “are to be awarded only in exceptional cases” that include “an especially egregious or outrageous matter.” A defendant can only be ordered to pay punitive damages if they engaged in “malicious” conduct or “acted in wanton and willful disregard” of the claimant’s rights.
You shouldn’t go into your claim expecting to receive punitive damages. Most claimants don’t receive punitive damages. But if your case progresses to trial and your doctor’s act of negligence was exceptional beyond that which is usually seen in a malpractice claim, punitive damages may be a possibility.
Aside from directly benefiting the claimant who receives this money, punitive damages can help to make a difference in the defendant’s future behavior. This expense may deter a doctor from engaging in subsequent negligent behavior and discourage hospital systems from continuing to utilize practices and procedures that put patient safety at risk.
When is it medical malpractice? To know if there’s a chance that you meet the medical malpractice requirements necessary for a claim, you should ask yourself two crucial questions:
If you answered yes to both questions – even if you’re not able to prove negligence at the moment – then your next step should be looking for a medical malpractice lawyer.
You see, your case is unique. The only way to find out if you have a claim is to talk to New Jersey medical malpractice lawyers with enough experience to know what to look for. There’s no simple answer key and no easy explanation that will tell you whether or not you have a case.
Why does determining if you have a case have to be so complicated?
In a complex medical malpractice claim, even the smallest details matter. That’s because these details are what serve as proof of the healthcare professional’s actions that fell below the standard of care.
No other patient has had the exact same doctor make the exact same mistake and suffered precisely the same injuries that changed their lives in precisely the same way. The facts involved in your medical ordeal are different from anyone else’s. These facts are what determine whether your case is actionable, and they’re what a lawyer will need to know before he or she can advise you about what to do next.
The challenge of winning a medical malpractice claim is that it isn’t malpractice just because you say it is. Every one of those medical malpractice elements – duty of care, breach of care, causation, and damages – must be backed up by evidence.
It takes significant evidence to show that your doctor acted negligently, that those negligent actions are what caused your injuries, and that your injuries are serious and permanent.
What might seem like common sense to you still requires expert testimony in a medical malpractice claim, particularly, in the eyes of the defendant’s insurance company. You should expect their lawyers to do everything they can to protect the hospital’s profits, even when the medical facility or provider is clearly in the wrong.
A lot of what determines your ability to pursue a medical malpractice claim is your prognosis. It takes a doctor to determine what your injuries are and what kind of recovery you can expect. That, in turn, affects all of your other damages – from the amount by which your future earning capacity is reduced to the extent of the injury’s effects on your quality of life.
The only way to prove negligence against a medical professional is through the expert opinion of another medical provider.
Remember, malpractice happens when the doctor deviates from the acceptable standard of care, not just when there’s an unwanted outcome – and it takes another doctor to say what’s acceptable.
Medical experts are credible doctors who have a great deal of experience practicing in a relevant area of medicine. They know their stuff – but that knowledge can come at a steep price.
It’s not unusual for a single malpractice case to involve multiple medical experts. When you have to pay each expert thousands of dollars for their services, the cost of your claim quickly becomes unaffordable.
It’s virtually always best to have a legal professional represent you in any legal matter. However, having an attorney is even more crucial in medical malpractice matters.
Despite the myths you might have heard, medical malpractice claims are far from slam-dunk cases. They require a lot of evidence in the form of expert opinions, and getting the right experts can be costly.
You’d like to think the law would be written to be fair to everyone. Unfortunately, that’s not the case. Often, medical malpractice laws are designed to protect doctors and hospitals, not patients. It’s bad enough that you can’t sue without severe injuries and an excessive amount of evidence. On top of that, the only way to prove that you were the victim of malpractice is through the testimony of experts – and they can be prohibitively expensive to attain.
Hiring a medical malpractice lawyer is your best chance of getting the results you need. Your attorney can be your advocate and work to level the playing field so that you have a fair chance at getting the money you deserve. And we can advance all upfront costs involved in pursuing your claim.
Can you win a medical malpractice suit? Yes, absolutely – but only when you combine the grounds for a viable claim with the skilled representation of a knowledgeable medical malpractice attorney.
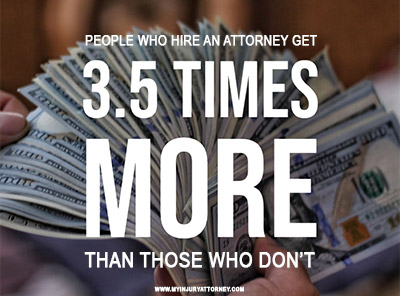 The biggest reason why you should always hire an attorney to represent you in medical malpractice suits is that it’s the only way to get the money you deserve.
The biggest reason why you should always hire an attorney to represent you in medical malpractice suits is that it’s the only way to get the money you deserve.
Data published by the Insurance Research Council has shown that attorneys get their clients 3.5 times more compensation than claimants without a lawyer. That means that even after you factor in the cost of hiring a medical malpractice lawyer, you’re ending up with a lot more money. You can better afford the costs of living with your injuries, of keeping your family afloat during tough financial times, and even of the most cutting-edge care that can help you recover.
In the case of a medical negligence situation, claimants have such a difficult time getting compensation to begin with that it is often not even possible, in this particular kind of claim, to get a successful outcome without an attorney.
But getting better results – or any results at all – isn’t the only reason to hire a New Jersey medical malpractice lawyer. Besides getting you a payout, your attorney will work hard to make your life after a medical error easier.
At the law firm of Console & Associates, we know that the aftermath of a medical error can be the most difficult time of your life. It’s our goal to not only get you the medical malpractice payout you deserve but also to make this time easier for your family.
Hiring an attorney offers you the best chance of getting the money you deserve for your malpractice claim, but it also helps you in so many other ways. With a lawyer on your side, you can pass the hassles of a legal matter onto us. That means 1) no more distractions from your rehabilitation and 2) someone always on your side. If you encounter problems getting or paying for care or you get bad news about the status of your job while you’re out of work, we’re here for you, no matter what.
Medical errors are alarmingly common, so much so that they constitute a leading cause of death in America.
Yet, due largely to the huge burden of evidence placed on the malpractice victim and the expense of moving forward with a claim, most instances of medical mistakes go unpunished. The victims don’t pursue a claim and never get the money they need to get life back on track. The negligent healthcare professionals get away with their dangerous treatment of patients, facing no consequences and free to do even more harm over the course of their medical career.
Historically, only 9 percent of civil trials have been medical malpractice claims, according to the United States Bureau of Justice Statistics. There were more than 3.5 times more motor vehicle accident trials than medical malpractice trials, even though – by some estimates – medical errors kill more than 12 times as many Americans as car accidents do. The reality is that victims of malpractice, and their families, are far less likely to get justice than victims of other types of accidents are.
The only way you can expect to get justice for medical malpractice is by hiring a lawyer. Hospitals and health systems, medical malpractice insurance carriers, and the massive and influential teams of defense attorneys on their side are just too powerful for one person to stand up to on their own.
One call to Console & Associates puts your claim in the hands of an accomplished multi-attorney firm, complete with experienced medical malpractice paralegals and a network of medical experts at our disposal. With us, you’ll have a full legal team on your side, without a hefty price tag to worry about.
Although it’s not advisable, it is possible to settle a personal injury claim, in general, yourself – but that’s not necessarily true for a medical negligence claim.
Claimants routinely settle minor, straightforward car accident claims on their own (although motor vehicle claims with more extensive damages should be handled by a lawyer). But pursuing a claim for medical malpractice without a lawyer is a very different situation. Medical malpractice claims are challenging and expensive to pursue because they require the use of medical experts and long hours of investigation.
Let’s compare handling car accident claims on your own to settling medical malpractice without a lawyer.
You don’t necessarily need expert witnesses for a car accident claim. Having them can add a lot of value, especially to a complicated claim. However, as a layperson, you can look up traffic safety laws and point out that the other driver was ticketed by the police for a violation. Although the cause of an accident can be a lot more complicated than that, there are also claims where proving negligence is relatively straightforward and an expert witness isn’t a necessity.
You do, however, need expert witnesses to pursue a matter of medical negligence. It’s the law.
Under New Jersey law, you aren’t qualified to pursue a medical malpractice lawsuit just because you, as a layperson, allege that malpractice occurred. It doesn’t matter if you have enough healthcare knowledge yourself to know that your doctor shouldn’t have done what they did. It doesn’t matter how many parts of your medical records you can point to that call into question your provider’s actions or omissions.
Under New Jersey statute §2A:53A-27, you must be able to provide an affidavit of lack of care to move forward with your medical malpractice lawsuit, according to the National Conference of State Legislatures. And, under New Jersey statute §2A:53A-41, only a licensed physician or other licensed healthcare professional is qualified to execute this affidavit or to provide the expert testimony on which the legal document is based. In addition to being licensed, the healthcare provider must meet strict criteria regarding the specialized certification and clinical practice to be authorized to make this sworn legal statement.
Because you must present this testimony and certify in advance that you have sufficient evidence to back up your claim through the affidavit of merit, the only way to move forward with your lawsuit is to have an expert witness on your side. Hiring an expert witness on your own could cost you thousands of dollars upfront – money you’d have to pay right now, whether or not you win your claim – and you’d still have no way to navigate the legal system.
You simply can’t handle this kind of claim yourself. You don’t have the experience or legal knowledge to do so. You also would have to spend a lot of your own money to secure the services of medical experts, which adds yet another hurdle to getting the compensation you deserve.
What you can do is choose a law firm like Console & Associates P.C. with a history of success in handling medical malpractice cases to represent you. Obtaining justice is a right that every victim, not only those with huge financial resources, should have.
Tens of thousands of dollars.
That’s how much it can cost just to gather the evidence you need to pursue your medical malpractice case.
But when you choose Console & Associates P.C.’s experienced New Jersey medical malpractice lawyers to handle your case, the astronomical cost doesn’t have to be your problem anymore.
If every medical malpractice victim had to come up with the kind of money necessary to pay numerous medical experts upfront, a lot of victims would never have the opportunity to get what they deserve. That’s not right or fair.
We believe that every victim harmed by medical negligence should have the right to hold the careless doctor accountable and receive the compensation they’re entitled to. So, when you become our client, we cover the upfront costs of pursuing your claim:
Only after we win money for you will we be reimbursed for these costs. You never have to worry about coming up with the cash on your own. And if we’re not able to get you compensation, then these costs remain our problem – never yours.
Securing the testimony of medical experts at no upfront cost is just one of the guarantees we make to you in our No Fee Promise. You shouldn’t have to risk thousands of dollars to get justice. You’ve already been through enough. We’re confident enough that we can win the cases we take on that we’re willing to take these risks for our clients.
A medical negligence case is costly, but medical malpractice help doesn’t have to be.
Don’t buy into the myth that you can’t afford an attorney for a medical malpractice claim in NJ. Here’s how you can get help from an experienced medical malpractice lawyer free of upfront charges – and only pay for legal representation if you win.
How do medical malpractice lawyers get paid? Most personal injury lawyers work on what’s called a contingency fee basis.
Under a contingency fee model, you pay your attorneys only a percentage of what they get for you. When you hire our New Jersey medical malpractice lawyers to handle your claim‚ you pay nothing out of pocket – from the initial consultation to negotiating with insurance adjusters, from hiring medical experts to your day in court (if it comes to that).
This means you only pay for legal representation…
You don’t have to pay now to speak to a medical malpractice attorney. The case evaluation and initial medical record review are free.
You don’t pay for calls, meetings, or every hour spent working on your case. No matter how many times you need to talk to your legal team, we won’t charge you for it. We’re at your disposal from the time you hire us to the time your case closes.
You won’t ever have to worry about racking up more in legal fees than your claim is worth. We collect only a percentage of what we win for you – and if we don’t win‚ you owe nothing. Since we get paid only a portion of what we get for you, it’s not possible that pursuing a medical malpractice lawsuit could backfire and end up costing you more money.
There’s one more benefit to working with New Jersey medical malpractice lawyers on a no-win, no-fee basis. A major reason why medical malpractice lawyers take claims on a contingency fee basis is that this practice aligns our goals with yours.
Think about it: no one else involved in your claim wants the same outcome you do. You, naturally, want to get the full amount of money you’re entitled to.
The negligent doctor doesn’t want that to happen. The hospital doesn’t want that. The insurance carriers that are actually paying out the claim don’t want that – they’d rather keep that money for themselves. The defense attorneys are actively fighting against you to prevent you from getting the money you deserve.
But the medical malpractice lawyer you hire on contingency? We have every incentive to work as hard as we need to just to get you every dollar you deserve because our pay is directly tied to how much money you recover.
If we didn’t charge medical malpractice lawyer fees on contingency, you wouldn’t have this reassurance.
Under a contingency fee agreement, you won’t have these worries. You can feel confident about your claim and the results our NJ medical malpractice attorneys get for it.
What percentage do medical malpractice lawyers get from a successful claim?
Under state law, New Jersey med mal lawyers can charge 33 1/3 percent of the first $750,000 of a settlement or jury award.
If your settlement or jury award exceeds $750,000, the percentage you pay on the remaining settlement amount (after collecting 33 1/3 on the first $750,000) decreases as follows:
Paying as much as a third of your settlement may sound like a lot. However, an attorney brings to your claim value far above what you’ll pay in attorneys’ fees.
The success of your medical malpractice claim hinges on the quality of your legal representation. How do you choose a good attorney who meets all of the most important medical malpractice lawyer qualifications?
Look for a lawyer who has:
These qualities are hard to judge when you don’t have a background in medical malpractice law yourself. Fortunately, there are factors you can look at that are more readily apparent and which shed some light on how well an attorney meets these requirements.
If you’re wondering who is the best medical malpractice lawyer, here are the top factors you need to consider:
What sets the medical malpractice attorneys of Console & Associates apart? We’re glad you asked.
This is why you want your medical malpractice claim in our capable hands:
Extensive experience in handling all areas of medical negligence claims.
We have more than 25 years of experience helping clients in New Jersey with their personal injury claims.
We’re fully prepared to hold negligent medical professionals accountable for mistakes committed in any environment, from in-home healthcare services to the state’s busiest emergency rooms.
Our record of success handling medical malpractice claims in NJ
We have helped thousands of clients with medical malpractice and other types of personal injury claims.
We have recovered numerous six-figure and seven-figure settlements on behalf of our clients, totaling well over $100 million.
Our exclusive focus on personal injury matters
Medical malpractice claims are too complex to trust to a general practice attorney. Our lawyers handle injury cases exclusively. We’re working in this complex area of law, and helping people in situations like yours, every single day.
Our familiarity with the hospital systems all over the state. Having practiced personal injury law throughout NJ for decades, we’re knowledgeable about the practices and reputations of the largest hospitals in the state, including:
Taking on a major medical system or a well-known doctor can be a scary prospect, but we won’t be intimidated. We’ll be here fighting for you every step of the way.
A Full Legal Team On Your Side
Different lawyers may look at the same case somewhat differently. One benefit of choosing Console & Associates P.C. to handle your claim is that we have three attorneys on staff. Often, our lawyers will each review your situation and discuss your legal matter as a team.
Don’t worry, it won’t take long. When we get back to you, you can be sure that a group of experienced attorneys has given your case the personalized attention it deserves.
In addition to our attorneys, your legal team includes knowledgeable paralegals and legal support staff who are always here to help. With us, you’re not just hiring a lawyer – you’re hiring our law firm in its entirety and taking full advantage of all we have to offer.
To find out if you can pursue a claim, you’ll need a lawyer on your side. Every case at Console & Associates begins with a medical malpractice lawyer’s free consultation.
Here’s what you can expect during the initial consultation.
Although we welcome new client communications by email contact form, our initial consultation usually begins with a phone call. We know your time is valuable, so we strive to gather crucial information now before you ever set foot in our office, so as not to waste any of your time. To that end, we will need to ask you some questions.
We know that a medical malpractice matter can be confusing and emotionally difficult. It’s okay if you don’t have all the answers off the top of your head if you don’t fully understand what happened, or even if you need some support to get through telling us your story. We’re here to help you.
Some aspects of your medical care or treatment, and your current medical troubles, may be private and difficult to discuss with a stranger. Rest assured that this is a confidential conversation between you and a representative of our firm.
You don’t have to worry about us blabbing to a well-known doctor or an influential health system that you’re thinking of suing. We won’t share private details about your situation with anyone who isn’t involved in evaluating your claim and, if you have the grounds for a case, with moving forward with that claim.
And as far as feeling embarrassed? Don’t. We’re professionals. There’s nothing we haven’t heard over our decades in practice. We handle matters involving serious injuries every day, so we’re used to helping clients with sensitive topics pertaining to their health. We want to get you on the path to feeling better.
A lot of the details that affect whether or not you have the grounds for a valid claim are found within your medical records. Because of this, reviewing your medical records is a crucial part of thoroughly evaluating your claim.
If, after speaking to you about your situation, we believe we may be able to help, we will ask your permission to review your medical records. You may not have your medical records yet at the time you speak with us. That’s okay. We can walk you through the process of getting your records or, in some instances, request those records for you. Our thorough review of your medical records allows us to consider all of the unique facts about your claim and determine whether there is sufficient evidence to prove that your healthcare provider was negligent.
Talking to an attorney should provide you with answers, not more questions. But that’s not always the case, especially when medical malpractice victims feel rushed or like they aren’t being listened to. Throughout your free case evaluation, you have our full attention. Besides asking you questions, we’ll help you understand what the legal process might look like if you pursue a claim.
A big part of educating and advocating for you is answering your questions. Whatever it is you’re wondering about, don’t hesitate to ask.
You’re not bothering us, and we won’t think your question is stupid. People come to us with varying levels of knowledge of law and medicine – both complicated topics – and we’re used to answering questions that range from the simple to the complex. What matters to us is that you have the knowledge you need to fully understand your situation, your options, and your legal rights.
Your medical malpractice claim may be your first time attempting to navigate the complicated legal process. Don’t worry. We handle malpractice and injury claims every single day, and we’ll be here to walk you through it from start to finish.
One of the biggest reasons malpractice victims don’t get the compensation they deserve is because they might be reluctant, or even afraid, to engage in the claims process. You don’t know what to expect, what pursuing a claim will require from you, or whether a medical malpractice claim could make things even worse.
Although winning malpractice cases is challenging, the legal process doesn’t have to be a hassle for the claimant. Once you choose a New Jersey medical malpractice lawyer to fight for you, we handle every aspect of your claim, so you don’t have to.
Here’s an overview of the medical malpractice lawsuit process:
Because there’s no risk and little hassle for you, the legal process of seeking money for a devastating medical mistake is often much easier on our clients than they expected. If worries about the process of filing a claim are what’s holding you back from getting the money you deserve, give us a call. Let us answer your questions and ease your mind.
When you move forward with a med mal claim, it’s not the doctor directly that you’re seeking compensation from. Instead, an insurance company is the one that will be paying out your claim.
Medical malpractice insurance is a type of professional liability insurance for healthcare providers. Generally, doctors in New Jersey must carry at least $1,000,000 in malpractice insurance under state law.
Other healthcare providers, like nurse practitioners and physician assistants, also carry malpractice insurance or professional liability insurance appropriate to their scope of practice. Hospitals and other types of medical facilities, too, purchase malpractice insurance. Healthcare facility staff members who do not have their own malpractice insurance policy may be covered by their employer’s or supervising physician’s policy.

There’s a lot you can gain from moving forward with your malpractice claim: answers, justice, accountability, a real reason to believe that the negligent doctor will avoid making the same mistake again. Ultimately, though, the direct goal of your medical error lawsuit is to secure your family financial compensation for your damages.
What is the average medical malpractice case worth? Many med mal claims end in a settlement or verdict in the six-figure or seven-figure range. The average payout amount in New Jersey is slightly higher than the national average.
The average medical malpractice payment in 2018 was $348,065, according to data collected by the National Practitioner Data Bank and presented by the doctor lending and insurance marketplace LeverageRX.
With 11,584 payments made in the United States for that year, the total payout amount for all medical malpractice claims reported was an astounding $4,031,987,700.
The vast majority – 96.5 percent – of these payouts came from out-of-court settlements, accounting for $3,889,417,950 in total payments. The 3.5 percent of medical malpractice payouts that were judgments culminated in a collective payment of $142,569,750.
The average amount of a medical malpractice payout can share some important information, but the value of your claim could be considerably more or considerably less. Remember, your medical malpractice matter is unique, and it depends on the damages and circumstances that pertain to your case.
Some medical malpractice claims have such limited damages that they are worth less than $100,000. Others bring in multimillion-dollar payouts.
In 2018, New Jersey was the state with the fifth-highest total medical malpractice payout amount, LeverageRX reported.
Over the course of the year, payments were made to 630 malpractice victims and their families totaling $226,712,000. The average payment in New Jersey was $359,860.
The real question isn’t just how much your case is worth, but whether the expected outcome will surpass the costs of bringing the claim.
How can you know if your injuries are serious enough for a successful medical malpractice case? After all, you certainly think they’re significant when you’re forced to struggle with them every day. But with the strict requirements, it can be hard for you to tell for sure.
Here’s a secret: without knowing the details of what happened to you, even experienced lawyers like us just don’t know if you have a case or not.
That’s why we need to understand all of the complicated forces at play in your claim. It’s why we need to review your damages and to hear – from medical experts and from you personally – what kind of damages you suffered because of the medical error.
According to New Jersey law, as of 2020, there is no limit on the damages you can seek to compensate you for a medical malpractice claim, also known as compensatory damages.
This means that your pain and suffering, lost wages, medical expenses, and ongoing care are all covered. There’s no cap on the amount of damages you could be awarded to compensate you for the harms you suffered.
Over the past decade, bills have been introduced at the state level that would aim to cap the amount of non-economic damages a medical malpractice victim could receive. For example, New Jersey Assembly Bill 966, proposed during the 2012-2013 state legislature session, would have capped payment for non-economic damages like pain and suffering at $250,000. Fortunately, that bill died in committee, preventing the government from infringing on the rights of medical malpractice victims to recover full compensation for their compensatory damages.
The state does, however, place a limit on punitive damages, which are only awarded in exceptional circumstances, not routinely. Under New Jersey statute 2A:15-5.14, you can only seek up to five times the award for your compensatory damages or $350,000, whichever is greater.
Your doctor’s medical error has changed your life forever. But your legal rights won’t last nearly as long as the damage their negligence has caused.
Your time to move forward with a claim is limited, and the clock is ticking. If you don’t take action before the statute of limitations passes, you may never end up getting the compensation you deserve.
What is the statute of limitations for a medical malpractice suit? Simply put, a statute of limitations is a regulation that restricts the time you have to pursue a legal matter.
Under New Jersey statute 2A:14-2, a medical malpractice claim generally must be brought within two years from the date of the incident. That’s in line with the statute of limitations for other types of personal injury actions.
Like other types of laws, statutes of limitations can be complicated. There are exceptions to this two-year period that applies to most situations.
Some situations could extend the deadline to file, and others could make that deadline much much earlier. To make sure you’re meeting all deadlines that apply to your claim, the sooner you contact an experienced attorney, the better.
When a car accident occurs, the incident is immediately apparent, even if not all of its consequences are. That isn’t always the case in a medical negligence matter.
You’re likely to notice a wrong-site surgery right away, but a foreign object left inside the body during surgery may take time to become apparent. Similarly, it’s not until time passes and an accurate diagnosis is made that you discover that a misdiagnosis or delayed diagnosis occurred.
In recognition of this fact, the discovery rule applies to medical negligence matters in NJ. Under the discovery rule, the two-year statute does not necessarily begin to run on the date the malpractice occurred. Instead, the statute of limitations for this type of med mal claim is two years from the date the person reasonably became aware that he or she had suffered malpractice.
If the medical error caused harm to a minor – defined as anyone under the age of 18 at the time of the incident – then a different set of deadlines applies.
The parents of a child harmed by malpractice can take action on the minor’s behalf right away. If the parents or guardians do not take action by the minor’s 12th birthday, NJ statute 2A:14-2 gives the minor the right to commence legal action on their own or to designate another adult to do so on their behalf.
Early on in your rehabilitation, it may seem like you have a while before the medical malpractice time limit becomes a problem. But you shouldn’t wait until the medical malpractice statute of limitations is looming before you decide to start filing a medical malpractice lawsuit. The sooner you seek legal help, the better.
An attorney can review your medical records to understand what went wrong – and that knowledge may be able to help your current practitioners fix it. We can make sure you’re getting the care you need and secure your medical records to make sure the evidence that supports your claim is preserved. From day one, we’ll gather and organize all of your medical bills and keep track of the time you’re out of work and the wages that you missed, so you won’t have to worry about anything slipping through the cracks.
From the day you choose us to handle your case, we’ve got your back. Why wouldn’t you want to get skilled, professional help on your side sooner rather than later?
Because Console & Associates bills on a contingency fee basis, there’s no monthly retainer or fee and no hourly charges. It doesn’t cost you anything to have legal representation lined up early on in your claim. Think of it as a safety net.
If you believe that you were a victim of medical malpractice – or that malpractice is behind the loss or catastrophic injury of your loved one – then it makes sense to speak with a New Jersey medical malpractice attorney immediately.
Don’t wait – call (856) 778-5500 today for your free case evaluation!
Just how common is medical malpractice and negligence? The data behind medical malpractice rates is nothing short of shocking.
Medical malpractice kills an astounding number of Americans each year – so much so that medical error is the third leading cause of death in the U.S., according to CNBC.
Only heart disease and cancer kill more people each year than medical mistakes do.
Research on medical malpractice by Johns Hopkins Medicine in 2016 found that medical errors kill more than 250,000 American patients annually.
A medical malpractice journal article published in the Journal of Patient Safety in 2013 suggested that as many as 400,000 premature deaths occur each year due to preventable harm to patients.
To further put these figures in perspective, here’s how fatal medical errors compare to firearm deaths. Every year, well over 30,000 Americans – often, closer to 40,000 Americans – lose their lives to gun violence.
At the most conservative estimates, medical errors kill 6 to 8 times more people than guns do in the United States.
For every patient who loses their life because of a doctor’s mistakes, many more suffer life-altering injuries that may require round-the-clock care.
The same Journal of Patient Safety article that put the likely number of fatalities due to medical errors at 400,000 per year also estimated that serious but non-lethal harm was “10- to 20-fold more common” than actual deaths. That means as many as 4 million to 8 million patients each year are maimed – sometimes irreparably – by negligent healthcare professionals.
Nearly 30 percent of medical malpractice payouts in 2018 resulted from a death due to medical error, according to LeverageRX. Although that makes death the single most common outcome to lead to a payout, it also means that, in more than two-thirds of all paid claims, patients were suing over non-fatal injuries.
Some deviations from the standard of care happen far more often than others. According to StatPearls Publishing, the nine most common medical errors in recent years have included, in order:
Other medical errors that occur too often include, in no set order:
These common medical mistakes can occur in all types of medical facilities and environments, but they tend to have the most serious consequences when they happen in:
Despite the massive number of harmful medical errors that occur each year in the United States, too many victims and their families never get the justice they deserve, because they don’t have the right help on their side.

Medical Malpractice Lawyer, Richard P. Console Jr.
When you’ve been harmed by a medical mistake, you have a lot to lose. There’s your health, your job and income, your family’s savings, and your quality of life.
But when you turn to the right lawyer to handle your medical malpractice claim, you take charge of the situation. Suddenly, you have a lot to gain: the money you deserve and the accountability of the doctor who hurt you. And, with our no-fee promise, you’ve got nothing to lose.
So why wait any longer to get help?
Remember, the consultation is always free. We’ll answer your questions about the legal process and investigate what went wrong with your medical care. And, with three convenient NJ locations plus a secure, fully remote intake process, we make it easy to sign up for the no-win, no-fee legal representation you need to get your life back.
Retaining an experienced medical malpractice lawyer can be as easy as making a phone call. Get our New Jersey medical malpractice lawyers on your side today!
You’ve got medical malpractice questions. We’ve got the information you need, always offered free of charge and from the reliable source of a law firm with more than 25 years of experience handling claims like yours.
A medical malpractice lawsuit is a civil, rather than criminal, legal matter. In certain instances, however, the acts that constitute medical malpractice also have a criminal component.
When is medical malpractice criminal?
This isn’t to suggest that facing criminal charges related to a malpractice matter is common. It’s not. However, there are plenty of possible scenarios in which a medical professional doing the wrong thing might be both negligent and criminal.
Doctors who are convicted of criminal charges may lose their license to practice medicine and face jail time and other criminal penalties. In this way, they will face the consequences of their actions, at least the criminal ones. However, you should still move forward with a claim against a doctor who is facing criminal charges.
Why? If you were harmed by their medical negligence, you have very real losses – financial and otherwise – for which you deserve compensation. Your doctor going to jail may be a moral victory, leaving you feeling vindicated and confident that the negligent medical professional won’t have the chance to harm anyone else. What won’t happen, unless you pursue a civil claim, are you acquiring the compensation you need to get your life back on track.
Not getting this money can prevent you from getting better and cause your family to struggle financially – possibly for years – due to someone else’s negligent actions. And that’s a risk you can’t afford to take.
Some claimants affected by medical malpractice close their cases too early. By the time they realize this is a mistake, it’s too late.
Perhaps you made an initial demand of the doctor or hospital and – to your surprise – the defendant agreed. This is most likely what we call a “nuisance” payment, made to get you to go away and get the consequences of the medical error to disappear. If this happened, it might be an indicator that you deserve a lot more for your claim. Otherwise, the other side wouldn’t have been so willing to pay.
Maybe you planned to pursue a claim but decided not to go through with it. If the statute of limitations has already passed on your legal matter, you’ve missed the deadline. It’s highly unlikely that any attorney, no matter how experienced, can help you.
If you trusted your medical malpractice claim to an inexperienced attorney or a general practice lawyer, you might have been disappointed in your settlement.
Whatever the situation, you’re wondering if it’s possible to reopen your medical negligence case. Unfortunately, once your case is closed, it’s closed for good.
When you accept a settlement, you typically sign a document called a release. This legally binding agreement releases the defendant from any obligations toward you pertaining to the claim.
It’s critical that you make sure you are getting the maximum amount of money you deserve before you agree to a resolution of your claim. You do this by hiring an experienced medical malpractice attorney to handle your claim from the get-go.
If you’re contemplating a settlement offer but you’re not sure it’s enough, take advantage of our free settlement offer review. We’ll take a look at the facts of your claim and help you figure out if the settlement amount on the table is fair or if you could be getting more money.
In normal, non-emergency situations, doctors are supposed to get your consent for any procedures they perform. Signing a consent form – after carefully reviewing it and discussing the procedure and concerns with your doctor – is the right thing to do when you’re about to undergo medical testing or treatment.
Only, in your case, things didn’t go as planned. A medical error occurred that left you seriously injured. Now you’re worried that signing that consent form could have waived your right to bring a medical malpractice case.
You didn’t ruin your claim by signing a consent form. What you consented to was the treatment or procedure. You certainly did not consent to your doctor making an egregious medical error.
Having signed a consent form won’t prevent you from pursuing a claim for reasons such as the following:
This situation becomes more complicated in certain situations. For example, infection is a risk that could occur in all surgical procedures, even if your doctor isn’t negligent, but it’s still possible to sue for an infection that can be traced back to negligent actions such as failing to sterilize medical tools.
If you aren’t sure whether the negative outcome you suffered was a result of medical malpractice or if it was a risk of the procedure that you consented to undertake, it’s time to talk to a lawyer. We can review your medical records and help clear things up, at no charge to you. From there, you can take a look at your legal options and decide what to do next.
Do medical malpractice payouts count as taxable income? The short answer is “(Usually) no.”
One reason victims of medical malpractice may not get the money they deserve is that they believe that taxes will claim most of their settlement, anyway. Why go through the trouble of pursuing a lawsuit if you won’t see any of the money?
The reality is that damages paid to compensate you for a physical injury aren’t taxable. This includes payments for your physical injuries (and the resulting medical bills) and for any emotional distress, like pain and suffering or mental anguish, they cause, according to the IRS.
Historically, the IRS has also considered payments for lost wages non-taxable when they are awarded as a result of a personal physical injury. That’s a difference from lost wage payments awarded in other legal matters, such as cases involving employment law or contract law.
Generally, the only type of payout for a legal matter involving a personal physical injury that the IRS considers taxable income is punitive damages. Since punitive damages aren’t meant to compensate you for your losses, they don’t fit under the category of compensatory damages and are instead counted as “Other Income” by the IRS.
The upshot of all this is that the payout that is awarded in most medical malpractice claims can’t be taxed. You get to keep that money, minus your attorneys’ fees and the costs of pursuing your claim, and use it as needed to move forward with your life.
Call Console and Associates P.C. today and take the first step to getting your life back on track.
This law firm is very professional, courteous, and they get the job done. I strongly recommend this law firm.
Rating: 5/5 ⭐⭐⭐⭐⭐
Amy M.
Read more reviews on Google!
